By Jim Fitch and Simeon Jaggernauth
Proceedings of the Joint Oil Analysis Program (JOAP), D.O.D., Annual Conference
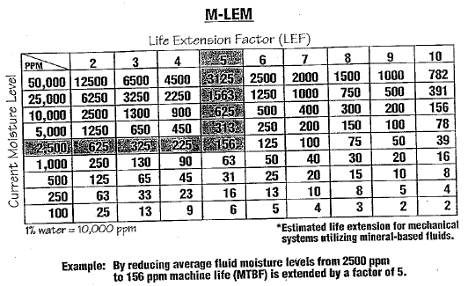
With few exceptions, the chemical and physical stability of lubricants are threatened by even the slightest amount of suspended water. Water can promote a host of chemical reactions (hydrolysis) with compounds and atomic species including oil additives, base stock and suspended contaminants. In combination with oxygen, heat, and metal catalysts, water is known to promote the oxidation and the formation of free radicals and peroxide compounds. Oxidation inhibitors are sacrificed by both neutralizing peroxides and breaking oxidation chain reactions to form stable compounds.
Other oxidation inhibitors are known to form hydrogen sulfide and sulfonic acids when reacting with water. Experiments have shown the protection provided by zinc dialkyldithio phosphate (ZDDP), a common antiwear additive and antioxidant, to be destroyed by as little as one drop of water in a gallon of oil, with oil temperature above l 80°F.
Water is also known to attack rust inhibitors, viscosity improvers, and the oil’s base stock. The effects are undesirable by-products such as varnish, sludge, organic and inorganic acids, surface deposits and lubricant thickening (polymerization). Large amounts of emulsified water can lower viscosity, thereby reducing a lubricant’s load carrying ability. When water is combined with metal catalysts such as iron or copper, accelerated stressing of the oil can occur. This results in base stock oxidation and the forming of free radicals (which continue the oxidation process), hydroperoxides, and acids (see figure 2).