By Jim Fitch & Simeon Jaggernauth
Proceedings of the Predictive Maintenance Technology National Conference
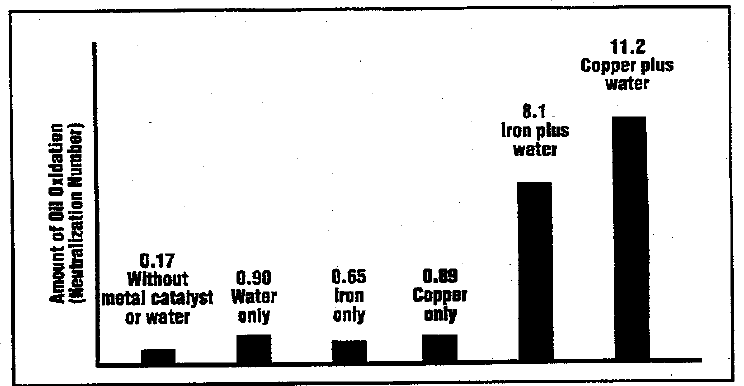
Water, once in an oil, is in constant search of a stable existence. Unlike oil, the water molecule is polar, which greatly limits its ability to dissolve; and many additives have polar extremities which can markedly increase water solubility. Water may cling to hydrophilic metal surfaces or form a thin film around polar solid contaminants such as silica particles. If a dry air boundary exists, water molecules may simply choose to migrate out of the oil to the far more absorbent air interface. This migration can be accelerated if air and oil mix, Such as in splash lubricated and oil mist systems or any system where a stable fluid foam may exist.
If water molecules are unable to find polar compounds on which to attach, the oil is said to be saturated. Any additional water will create a supersaturated condition causing free water to be suspended or settle at the bottom of the sump. This supersaturation can also occur as a result of lower oil temperature.